 Lithium ion batteries have become a key component of portable electronic devices as they offer high energy density, flexible lightweight design and a longer cycle life than other battery systems. More efficient batteries are required in the development of advanced transportation technologies in order to reduce the use of imported oil and the emission of greenhouse gas. Electrochemical energy storage has been identified as a critical enabling technology for advanced, fuel-efficient, light and heavy duty vehicles. New materials need to be designed to achieve higher energy/power densities, longer cycle lives and better reliability for such applications.
Lithium ion batteries have become a key component of portable electronic devices as they offer high energy density, flexible lightweight design and a longer cycle life than other battery systems. More efficient batteries are required in the development of advanced transportation technologies in order to reduce the use of imported oil and the emission of greenhouse gas. Electrochemical energy storage has been identified as a critical enabling technology for advanced, fuel-efficient, light and heavy duty vehicles. New materials need to be designed to achieve higher energy/power densities, longer cycle lives and better reliability for such applications.
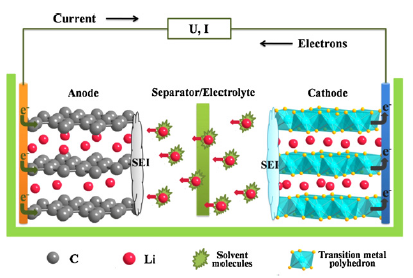
Cathode
To enable LIB as the main on-board storage technology in plug-in hybrid electric vehicles (PHEVs) or electric vehicles (EVs), higher energy density materials such as the ‘‘Li-excess’’ layered oxides, formed as the composites between Li[Li1/3Mn2/3]O2 and LiMO2 (M = Ni, Mn, Co), are promising candidates as they offer much higher capacity (>250 mAh/g) and better safety with much reduced cost. Currently, the large first cycle irreversible capacity, low rate capacity and votage/capacity fading are the major problems in this series of materials. We are developing various characterization techniques (x-ray, neutron, electron based in-situ/ ex-situ techniques) and modeling methods (VASP, FEFF, Wen2k) to study the mechanism of these problems.
Anode
After nearly three and a half decades of optimization, the energy density (Wh kg-1) of Li-ion batteries is quickly approaching the intercalation chemistry’s upper limit threshold. Before transitioning to a totally new chemistry, one final improvement slated for commercial batteries involves replacing graphitic anodes with Si. Si’s theoretical capacity (Ah kg-1) is an order of magnitude larger than that of graphite’s, which translates to a final overall battery energy density increase of approximately 10 – 15%. Unfortunately, Si anodes are not yet ready for commercialization because designing reversible Si electrode structures has presented researchers with formidable engineering challenges.
The large irreversibility associated with the formative cycles of Si anodes is primarily due to mechanical pulverization and chemical instability. The mechanical pulverization of the Si active material has largely been solved by adopting nanostructures. Additionally, a wide range of electronically conductive binders and backend electrode coatings help to address the pulverization of the overall electrode structure. It is not yet clear how to create a stable solid electrolyte interphase (SEI) between Si and the organic liquid electrolyte in order to improve electrode Coulombic efficiency and reversibility.
Our group is interested in characterizing the composition of the SEI formed on amorphous-Si thin film electrodes fabricated by pulsed laser deposition, nano particles, and porous Si.
Highlighted Publications
K. Kang, Y.S. Meng, J. Breger, C.P. Grey and G. Ceder, “Electrodes with high power and high capacity for rechargeable lithium batteries“, Science, 2006, 311, 977
B. Xu, C. R. Fell, M. Chi, and Y. S. Meng, “Identifying surface structural changes in layered Li-excess nickel manganese oxides in high voltage lithium ion batteries: A joint experimental and theoretical study“, Energy & Environmental Science, 2011, 4, 2223
D. Qian, Y. Hinuma, H. Chen, L-S Du, K. Carroll, G. Ceder, C. P. Grey, Y. S. Meng, “Electronic spin transition in nanosize stoichiometric lithium cobalt oxide“, Journal of the American Chemical Society, 2012, 134(14), 6096
B. Xu, D. Qian, Z. Wang, Y. S. Meng, “Recent Progress in Cathode Materials Research for Advanced Lithium Ion Batteries“, Materials Science and Engineering R, 2012, 73(5), 51
Y. S. Meng, M. E. A.-d. Dompablo, “Recent Advances in First Principles Computatinal Research of Cathode Materials for Lithium-Ion Batteries“, Accounts of Chemical Research, 2012, 46(5), 1171
